WILL THEIR NALOXONE
BE ENOUGH?
To prevent the detrimental effects of hypoxia during an opioid overdose emergency, it’s
critical that patients receive a timely and adequate dose of naloxone.1
When every moment matters during an opioid overdose emergency rescue situation
WILL THEIR
NALOXONE BE
ENOUGH?
To prevent the detrimental effects of hypoxia during an opioid overdose emergency, it’s critical that patients receive a timely and adequate dose of naloxone.1



equivalents per day).6 Learn more here.
risk of an overdose, such as higher
opioid dosages (≥50 morphine milligram
equivalents per day).6 Learn more here.
ZIMHI HELPS ENSURE YOUR PATIENTS ARE PREPARED
FOR AN OPIOID OVERDOSE SITUATION
One 5-mg intramuscular dose provides a higher plasma concentration and exposure compared with a 2-mg intramuscular dose7
Mean plasma concentration of naloxone over 30 minutes
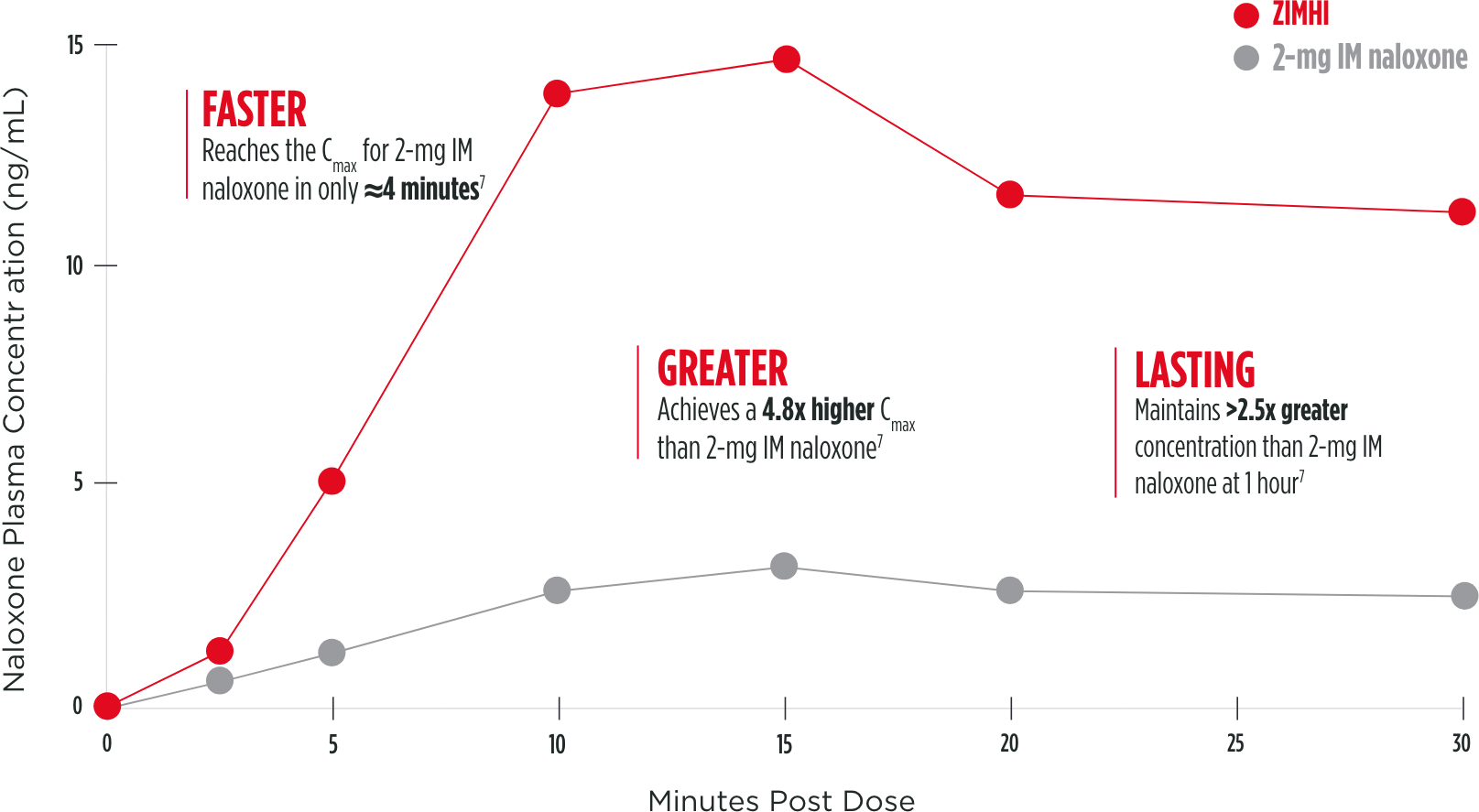
Note: The clinical relevance of these pharmacokinetic data is unknown. These data do not account for interpatient variability.
Cmax=maximum concentration; IM=intramuscular.
ZIMHI HELPS ENSURE YOUR PATIENTS ARE PREPARED FOR AN OPIOID OVERDOSE SITUATION
One 5-mg intramuscular dose provides a higher plasma concentration and exposure compared with a 2-mg intramuscular dose7
Mean plasma concentration of
naloxone over 30 minutes
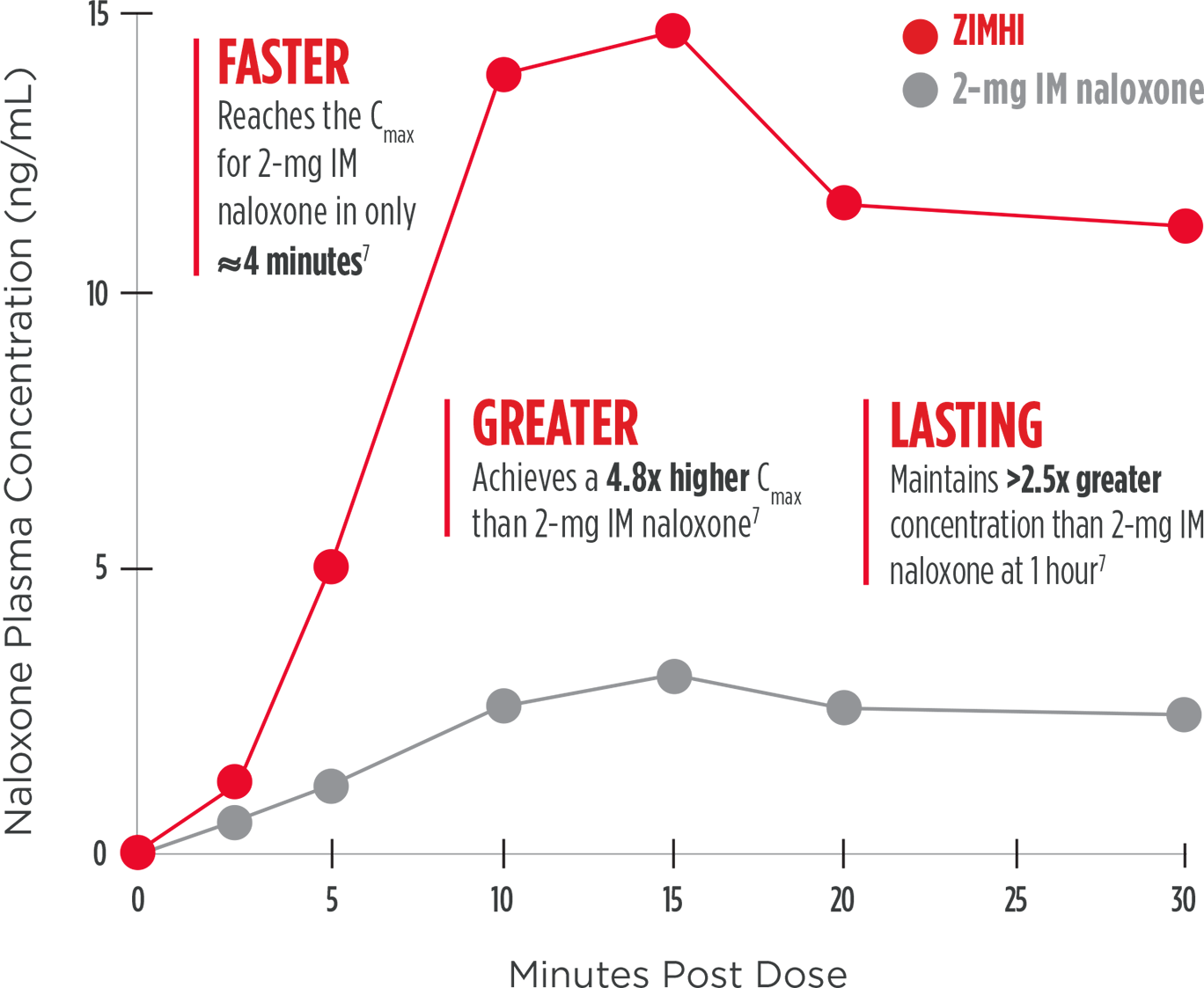
Note: The clinical relevance of these
pharmacokinetic data is unknown. These data
do not account for interpatient variability.
Cmax=maximum concentration; IM=intramuscular.

EVERY MOMENT MATTERS
The strength to pull them back from an overdose
REVIEW THE 4 SIMPLE STEPS FOR USE
HELPFUL TIPS FOR TALKING TO
PATIENTS ABOUT ZIMHI
ORDER ZIMHI

EVERY
MOMENT
MATTERS
The strength to pull them
back from an overdose
REVIEW THE 4 SIMPLE STEPS FOR USE
HELPFUL TIPS FOR TALKING TO
PATIENTS ABOUT ZIMHI
ORDER ZIMHI

EVERY
MOMENT
MATTERS
The strength to pull them
back from an overdose
REVIEW THE 4 SIMPLE STEPS FOR USE
HELPFUL TIPS FOR TALKING TO
PATIENTS ABOUT ZIMHI
ORDER ZIMHI
In an opioid overdose emergency,
ZIMHI MAY MAKE ALL
THE DIFFERENCE


In an opioid overdose emergency,
ZIMHI MAY
MAKE ALL THE
DIFFERENCE


In an opioid overdose emergency,
ZIMHI MAY
MAKE ALL THE
DIFFERENCE
References: 1. FDA Advisory Committee on the most appropriate dose or doses of naloxone to reverse the effects of life-threatening opioid overdose in the community settings. Adapt Pharma Operations Limited; 2016. 2. Spinal Cord. How long can the brain go without oxygen? What happens? Accessed November 30, 2021. https://www.spinalcord.com/blog/what-happens-to-the-brain-after-a-lack-of-oxygen. Spinal Cord, Inc.; 2019. 3. Family Caregiver Alliance. Hypoxic-anoxic brain injury. Published 2004. Accessed November 30, 2021. https://www.caregiver.org/hypoxic-anoxic-brain-injury 4. Medline Plus. Opioid overdose. Accessed October 28, 2020. https://medlineplus.gov/opioidoverdose.html Updated October 8, 2020. 5. Mell HK, Mumma SN, Hiestand B, Carr BG, Holland T, Stopyra J. Emergency medical services response times in rural, suburban, and urban areas. JAMA Surg. 2017;152(10):983-984. doi:10.1001/jamasurg.2017.2230 6. Centers for Disease Control and Prevention. Calculating total daily dose of opioids for safer dosage. Accessed November 30, 2021. https://www.cdc.gov/drugoverdose/pdf/calculating_total_daily_dose-a.pdf 7. ZIMHI. Prescribing information.
INDICATION
ZIMHI is an opioid antagonist indicated for the emergency treatment of known or suspected opioid overdose, as manifested by respiratory and/or central nervous system depression in adult and pediatric patients. ZIMHI is intended for immediate administration as emergency therapy in settings where opioids may be present. ZIMHI is not a substitute for emergency medical care.
IMPORTANT SAFETY INFORMATION
As the duration of action of naloxone hydrochloride is shorter than many opioids, keep the patient under continued surveillance and administer repeated doses of naloxone using a new ZIMHI device, as necessary, while awaiting emergency medical assistance.
Reversal of respiratory depression caused by partial agonists or mixed agonists/antagonists, such as buprenorphine and pentazocine may be incomplete. Repeat doses of ZIMHI may be required.
Use in patients who are opioid dependent may precipitate opioid withdrawal. In neonates, opioid withdrawal may be life-threatening if not recognized and properly treated. Monitor for the development of signs and symptoms of opioid withdrawal.
Abrupt postoperative reversal of opioid depression may result in adverse cardiovascular (CV) effects. These events have primarily occurred in patients who had pre-existing CV disorders or received other drugs that may have similar adverse CV effects. Monitor these patients closely in an appropriate healthcare setting after use of naloxone hydrochloride.
After use, the ZIMHI needle is exposed until the safety guard is deployed. A needlestick injury could occur during use in emergency situations. In the event of accidental needlestick injury, medical attention should be sought.
The following adverse reactions were most commonly observed in ZIMHI clinical studies: dizziness, lightheadedness, and elevated bilirubin.
To report SUSPECTED ADVERSE REACTIONS, call 1-800-230-3935 or FDA at
1-800-FDA-1088 or www.fda.gov/medwatch
IMPORTANT SAFETY INFORMATION and INDICATION for ZIMHI™
As the duration of action of naloxone hydrochloride is shorter than many opioids, keep the patient under continued surveillance and administer repeated doses of naloxone using a new ZIMHI device, as necessary, while awaiting emergency medical assistance.
Reversal of respiratory depression caused by partial agonists or mixed agonists/antagonists, such as buprenorphine and pentazocine may be incomplete. Repeat doses of ZIMHI may be required.
IMPORTANT SAFETY INFORMATION and INDICATION for ZIMHI™



